- English
- Español
- Português
- русский
- Français
- 日本語
- Deutsch
- tiếng Việt
- Italiano
- Nederlands
- ภาษาไทย
- Polski
- 한국어
- Svenska
- magyar
- Malay
- বাংলা ভাষার
- Dansk
- Suomi
- हिन्दी
- Pilipino
- Türkçe
- Gaeilge
- العربية
- Indonesia
- Norsk
- تمل
- český
- ελληνικά
- український
- Javanese
- فارسی
- தமிழ்
- తెలుగు
- नेपाली
- Burmese
- български
- ລາວ
- Latine
- Қазақша
- Euskal
- Azərbaycan
- Slovenský jazyk
- Македонски
- Lietuvos
- Eesti Keel
- Română
- Slovenski
- मराठी
- Srpski језик
Rocuronium Bromide API: How Does a Single Molecule Support General Anesthesia Worldwide?
Every day, in operating rooms across the globe, anesthesiologists perform a precisely choreographed sequence. The patient receives an induction agent to lose consciousness. Then comes a neuromuscular blocking agent—a drug that paralyzes skeletal muscles, including those required for breathing. The anesthesiologist inserts an endotracheal tube, connects a ventilator, and the surgery proceeds. Without the neuromuscular blocker, intubation would be impossible in a conscious or lightly sedated patient. Without the reversal agent, paralysis would outlast the procedure.
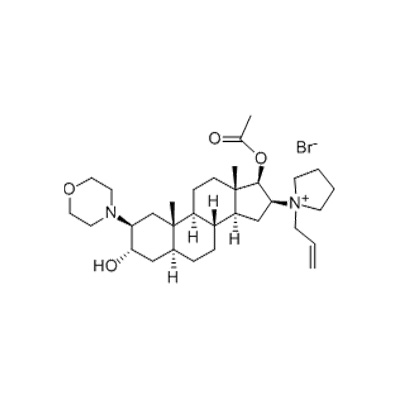
At the center of this sequence sits Rocuronium bromide, a non-depolarizing neuromuscular blocking agent with a molecular formula of C₃₂H₅₃BrN₂O₄ and a molecular weight of 609.67. This white to off-white crystalline powder, identified by CAS number 119302-91-9, is classified chemically as L-[(2β,3α,5α,16β,17β)-17-(acetoxy)-3-hydroxy-2-(4-morpholinyl)androst-16-yl]-1-propene pyrrolammonium bromide. In clinical terms, it is one of the most widely used muscle relaxants in modern anesthesia.
The Clinical Role of Rocuronium Bromide
Rocuronium bromide belongs to the aminosteroid class of neuromuscular blocking agents. It works by competitively blocking nicotinic acetylcholine receptors at the motor endplate of skeletal muscle. When the receptor is occupied by rocuronium, acetylcholine cannot trigger muscle contraction. The result is complete flaccid paralysis, beginning with small muscles of the eyes and face, progressing to the limbs, and finally affecting the diaphragm and intercostal muscles required for breathing.
Three characteristics have made rocuronium bromide a preferred agent in modern anesthesia:
Rapid onset of action : At intubating doses, rocuronium produces acceptable intubating conditions within 60 to 90 seconds. This speed approaches that of succinylcholine—historically the only agent capable of rapid sequence intubation—without succinylcholine's side effect profile of hyperkalemia, malignant hyperthermia, and muscle fasciculations.
Intermediate duration of action : A standard intubating dose provides paralysis for approximately 30 to 60 minutes, appropriate for most surgical procedures. Spontaneous recovery occurs as the drug redistributes and is eliminated, primarily through hepatic uptake and biliary excretion.
Reversibility with sugammadex : Unlike older neuromuscular blockers, rocuronium can be rapidly reversed at any time after administration using sugammadex, a modified gamma-cyclodextrin that encapsulates rocuronium molecules and renders them inactive. This allows anesthesiologists to reverse paralysis immediately after surgery rather than waiting for spontaneous recovery.
The availability of high-purity rocuronium bromide API directly affects the safety of millions of surgical procedures annually. Impurities or incorrect potency in the active ingredient could lead to inadequate paralysis—resulting in patient movement during surgery—or excessive paralysis—prolonging mechanical ventilation beyond the intended duration.
Manufacturing Infrastructure for Sterile API Production
Jiangsu Run'an Pharmaceutical's facility represents a significant investment in pharmaceutical manufacturing infrastructure. The 25,000 square meter building area houses production lines dedicated to fertility regulation drugs and variety APIs including rocuronium bromide.
Several aspects of the facility are particularly relevant to neuromuscular blocker API production:
Containment and cross-contamination prevention : Neuromuscular blocking agents are pharmacologically potent at milligram and even microgram doses. Cross-contamination of other drug products with trace amounts of rocuronium could cause unintended paralysis. The facility must therefore incorporate dedicated production areas, segregated HVAC systems, and validated cleaning procedures.
Analytical capabilities : Rocuronium bromide API requires testing for identity, purity, potency, and related substances. Analytical methods including high-performance liquid chromatography (HPLC) and mass spectrometry must be qualified and validated. The facility's 160 million yuan investment presumably includes these analytical capabilities.
Regulatory compliance : APIs for injectable neuromuscular blockers must meet pharmacopeial standards—typically United States Pharmacopeia (USP), European Pharmacopoeia (Ph. Eur.), or Chinese Pharmacopoeia requirements. The facility's design should accommodate the documentation, sampling, and testing protocols required for regulatory inspection.
The company states its aim "to become a world-class developer and manufacturer of reproductive products through constant improvement and innovation in technologies, and offer remarkable business opportunities to partner companies worldwide." For rocuronium bromide API specifically, this requires not only synthetic capability but also regulatory engagement with health authorities in target markets.
The API Portfolio Beyond Rocuronium
While rocuronium bromide is one of the better-known molecules in Jiangsu Run'an's portfolio, the company manufactures a substantial range of active pharmaceutical ingredients:
| API | Primary Therapeutic Use |
|---|---|
| Gemcitabine hydrochloride | Anticancer (pancreatic, lung, breast, ovarian) |
| Celecoxib | Nonsteroidal anti-inflammatory (arthritis, pain) |
| Bromhexine hydrochloride | Mucolytic (respiratory conditions) |
| Iguratimod | Disease-modifying antirheumatic drug |
| Apremilast | Phosphodiesterase 4 inhibitor (psoriatic arthritis) |
| Tofacitinib citrate | Janus kinase inhibitor (rheumatoid arthritis) |
| Crisaborole | Phosphodiesterase 4 inhibitor (atopic dermatitis) |
| Urapidil hydrochloride | Antihypertensive |
| Sugammadex sodium | Reversal agent for rocuronium and vecuronium |
| Garlicin | Cardiovascular (allium sativum extract) |
| Dexmedetomidine hydrochloride | Sedative (ICU and procedural sedation) |
| Rocuronium bromide | Neuromuscular blocker (anesthesia) |
| Finerenone | Nonsteroidal mineralocorticoid receptor antagonist |
| Sodium risedronate | Bisphosphonate (osteoporosis) |
This portfolio spans multiple therapeutic categories: oncology, rheumatology, anesthesia, critical care, dermatology, and metabolic bone disease. For a company founded on fertility regulation drugs, the expansion into neuromuscular blockers like rocuronium bromide represents strategic diversification.
The Rocuronium-Sugammadex Pairing
One of the most significant developments in modern anesthesia has been the clinical introduction of sugammadex. Before sugammadex, reversal of rocuronium-induced paralysis relied on acetylcholinesterase inhibitors (neostigmine) given with an anticholinergic agent (glycopyrrolate or atropine). This approach was indirect: increasing acetylcholine concentration to compete with rocuronium at the receptor.
Sugammadex works directly. It is a modified gamma-cyclodextrin with a lipophilic core that encapsulates rocuronium molecules, forming a stable complex that is excreted unchanged in urine. The effect is rapid—typically within two to three minutes—and predictable regardless of the depth of paralysis at the time of administration.
For API manufacturers, the pairing of rocuronium bromide and sugammadex sodium creates a co-dependent market. Hospitals using rocuronium prefer to stock its specific reversal agent. Jiangsu Run'an's portfolio includes both APIs, positioning the company to supply the complete neuromuscular blockade and reversal system.
Quality Attributes of Rocuronium Bromide API
For any pharmaceutical company sourcing or manufacturing rocuronium bromide API, specific quality attributes determine suitability for injectable formulation:
Appearance : White to off-white crystalline powder. Color deviations may indicate impurities or degradation.
Assay : Typically specified as 98.0% to 102.0% on anhydrous basis. Potency outside this range requires reformulation or rejection.
Related substances : Individual unspecified impurities limited to 0.1% or lower; total impurities constrained to specified limits. Rocuronium bromide has several potential degradation products and synthetic intermediates that must be controlled.
Residual solvents : Class 1 solvents (benzene, carbon tetrachloride, etc.) prohibited; Class 2 solvents limited per ICH Q3C guidelines.
Heavy metals : Limits for catalytic metals such as palladium or platinum if used in synthesis; general heavy metals limit typically below 20 ppm.
Microbial limits : For non-sterile API, limits for total aerobic microbial count, total combined yeasts/molds, and absence of specified pathogens (E. coli, S. aureus, P. aeruginosa, etc.).
Particle size : May be specified if the API is used in suspension or influences downstream processing.
The chemical structure of rocuronium bromide—a steroid backbone with a morpholinyl group and a pyrrolidinium quaternary ammonium—presents specific analytical challenges. The molecule has multiple chiral centers; stereochemical purity must be controlled, as isomers may have different pharmacological activity or safety profiles.
Strategic Positioning in the Global API Market
China has emerged as a dominant force in global API manufacturing, and Jiangsu Run'an represents the segment of that industry focused on higher-complexity molecules. Rocuronium bromide is not a simple commodity API. Its synthesis requires multiple steps, controlled reaction conditions, and purification processes that demand technical expertise.
For international pharmaceutical companies seeking API suppliers, several factors influence the evaluation of Jiangsu Run'an:
Parent company backing : As a wholly-owned subsidiary of Jiangsu Chiatai Qingjiang Pharmaceutical Co., Ltd., Jiangsu Run'an benefits from established quality systems, regulatory relationships, and financial stability.
Dedicated facility : The 2018 construction date means the facility was built under current good manufacturing practice standards rather than adapted from legacy infrastructure. This is advantageous for regulatory inspections by the US Food and Drug Administration, European Medicines Agency, or other authorities.
Portfolio depth : The range of APIs—from gemcitabine to rocuronium to sugammadex—suggests synthetic chemistry capability across multiple reaction types, including steroidal modifications and heterocyclic chemistry.
The company's stated commitment to "outstanding customer service" and "solid relationships with partners all over the world" reflects an understanding that API supply is not merely transactional. Pharmaceutical companies need technical support, regulatory documentation, supply chain reliability, and long-term partnership—not just a certificate of analysis.
Key Takeaways
- Active pharmaceutical ingredient: Rocuronium bromide (CAS 119302-91-9) — neuromuscular blocking agent for general anesthesia
- Molecular profile: C₃₂H₅₃BrN₂O₄, molecular weight 609.67, white to off-white crystalline powder
- Clinical role: Facilitates endotracheal intubation and provides surgical muscle relaxation; reversible with sugammadex
- Manufacturer: Jiangsu Run'an Pharmaceutical Co., Ltd. — wholly-owned subsidiary of Jiangsu Chiatai Qingjiang Pharmaceutical
- Facility: 59 mu (3.9 hectares), 160 million yuan investment, 25,000 m² building area, construction began November 2018
- API portfolio: Gemcitabine, celecoxib, bromhexine, iguratimod, apremilast, tofacitinib, crisaborole, urapidil, sugammadex, garlicin, dexmedetomidine, rocuronium, finerenone, risedronate
- Strategic focus: Fertility regulation drugs and variety APIs, aiming for world-class status in reproductive products
- Commercial positioning: Supplier relationships with international pharmaceutical partners; customer service commitment